Background and Objectives: Direct pharmaceutical marketing to physicians by pharmaceutical representatives is effective in changing behavior of health care providers, resulting in less evidence-based prescribing. Although much has been written about pharmaceutical marketing exposures among medical students, less is known about direct marketing exposures before students matriculate. This study examined the types of pharmaceutical representative direct marketing exposures for premedical students and where they occurred.
Methods: From June to August of 2017, researchers surveyed students who accepted admission to US public medical schools. These prematriculated students completed our survey just prior to matriculation. The survey inquired about whether the students were exposed to pharmaceutical marketing directly from pharmaceutical salespeople, the types of marketing they observed or received, and where these interactions occurred.
Results: Survey participants included 911 prematriculated students from 14 of the 188 medical schools invited to participate. Seventy-one percent (646) of the participants received or observed someone receiving pharmaceutical marketing gifts, small meals or snacks, articles, or samples. The two most common contexts for direct pharmaceutical marketing exposures were during shadowing experiences (54%, 346) and during employment (50%, 323).
Conclusions: The findings suggest that it may be common for medical students to have interacted directly with pharmaceutical salespeople or observed other health professionals in these interactions before they matriculate in medical school. Because many of these interactions occur during clinical experiences required by institutions for admission, medical schools and premedical associations should consider delivering conflict-of-interest education early in medical school education or before students matriculate.
Are the multinational producers of pharmaceuticals taking a page from big tobacco’s playbook? In the 1990s, it was demonstrated that the Joe Camel image in tobacco advertisements appealed to children more than adults, illustrating the seduction of marketing to younger, less experienced consumers.1 Similar to this marketing strategy used by the tobacco industry, pharmaceutical companies began interacting with medical students in formative stages of their careers.2-5
Direct marketing of pharmaceuticals to physicians by drug salespeople is a long-standing and controversial practice in medicine going back more than half a century.6 This type of marketing stands in contrast to pharmaceutical advertising to consumers through mass media. Direct marketing tactics frequently include pharmaceutical salespeople offering physicians and trainees small gifts, articles, small meals or snacks, or samples as a means to create an obligation among the recipients to repay these favors.7,8 These tactics have been shown to be effective in changing the behavior of both health care providers and physicians-in-training in ways that deviate from evidence-based practices. The result is less evidence-based prescribing behavior, decreased prescribing of generic alternatives, higher prescribing costs, and requests for formulary additions with little or no benefit over existing therapeutics.9-16
Progress has been made over the last 50 years. There is a lively discussion among medical ethicists, educators, and the public around this conflict of interest. Fewer physicians are interacting with pharmaceutical representatives this decade compared to last.12-17 Similar trends are observed with training programs.18,19
Despite the positive changes, the impact of the pharmaceutical industry-physician relationship continues to be felt. A 2012 Pew Foundation report found that the pharmaceutical industry is spending eight times more on direct pharmaceutical marketing to clinicians compared to marketing to consumers through mass media. There are an estimated 72,000 pharmaceutical representatives in the United States alone.20 Generalists, on average, receive two visits per day from pharmaceutical sales representatives.21
Medical students, faculty, and national organizations have all called for professionalism education around the physician-pharmaceutical industry relationship.22-26 As a result, in many schools medical students receive professionalism training that includes managing the physician-pharmaceutical industry relationship. However, if learners are exposed to direct pharmaceutical marketing before they even reach medical school, these efforts might be too late. Additionally, premedical student exposure to pharmaceutical marketing risks normalization of a conflict of interest known to lead to prescribing patterns that deviate from evidence-based practices. Students who feel that physician exposure to pharmaceutical marketing is acceptable or beneficial may sustain such exposure in their future training and practice despite its well-known harms.
To our knowledge, there has been only one study that has examined the context and prevalence of direct pharmaceutical marketing exposure among premedical students. Hodges et al found widespread exposure to pharmaceutical marketing among its students prior to entry into medical school. Additionally, they found shadowing was a common occasion for premedical students to interact with or observe pharmaceutical sales representatives. That study, however, was limited to a single school and was at high risk of recall bias because it surveyed students after matriculation into medical school, and in some cases in their fourth year of medical school.27 Our objective was to conduct a national study to further validate past findings of this localized study and further explore the prevalence and context of premedical students’ exposure to marketing delivered directly from pharmaceutical salespeople.
Participants
From March to June of 2017, our research group contacted deans of admissions or admissions administrators at all public medical schools in the United States requesting permission to survey their students who accepted admission to the entering 2017 class who had not yet matriculated at their institution. Study details and invitations to admissions deans to participate were issued by email. Follow-up phone calls were made to institutions that did not initially respond.
Those medical schools that chose to participate sent a scripted email invitation that our research team developed to their prematriculated medical students, inviting them to complete an online survey. Most participating institutions sent out reminder emails to participate. Student participation was voluntary and anonymous. Participants were encouraged to contact our research team if they had questions about the study.
Survey
Survey questions were based on a literature review and our previous research experiences in this area of inquiry.27-33 The survey queried the extent to which prematriculated medical students observed or received direct pharmaceutical marketing from a salesperson. Those who answered yes to any type of observed or received marketing interaction were asked a follow-up question about the context in which this interaction occurred (Table 1). We piloted the survey with two current medical students at the University of Washington for clarity, ease of use, and user acceptability.
Data Collection and Analysis
Data was collected from June to August 2017. We used a secure online web-based survey to collect student responses (UW Catalyst). Descriptive statistics were computed. An error in the survey logic prevented capture of student responses to one of the survey questions, “observing someone receiving a gift.” This question was excluded from our analysis.
The University of Washington Human Subjects Division reviewed this study and gave it exempt status.
Among the 188 pubic medical schools invited, 14 chose to participate (7% response rate). Table 1 shows the survey questions asked to each participant. Nine hundred eleven of 2,285 (40%) prematriculated students at participating institutions responded to the survey (Table 2). The mean age of the prematriculated participants was 25 years with a range of 21 to 44 years; 47% were female. Participating institutions were located in all four US census regions: three medical schools (302 participants) from the South; seven medical schools (342 participants) from the Midwest; one medical school (66 participants) from the Northeast; and two medical schools (200 participants) from the West.
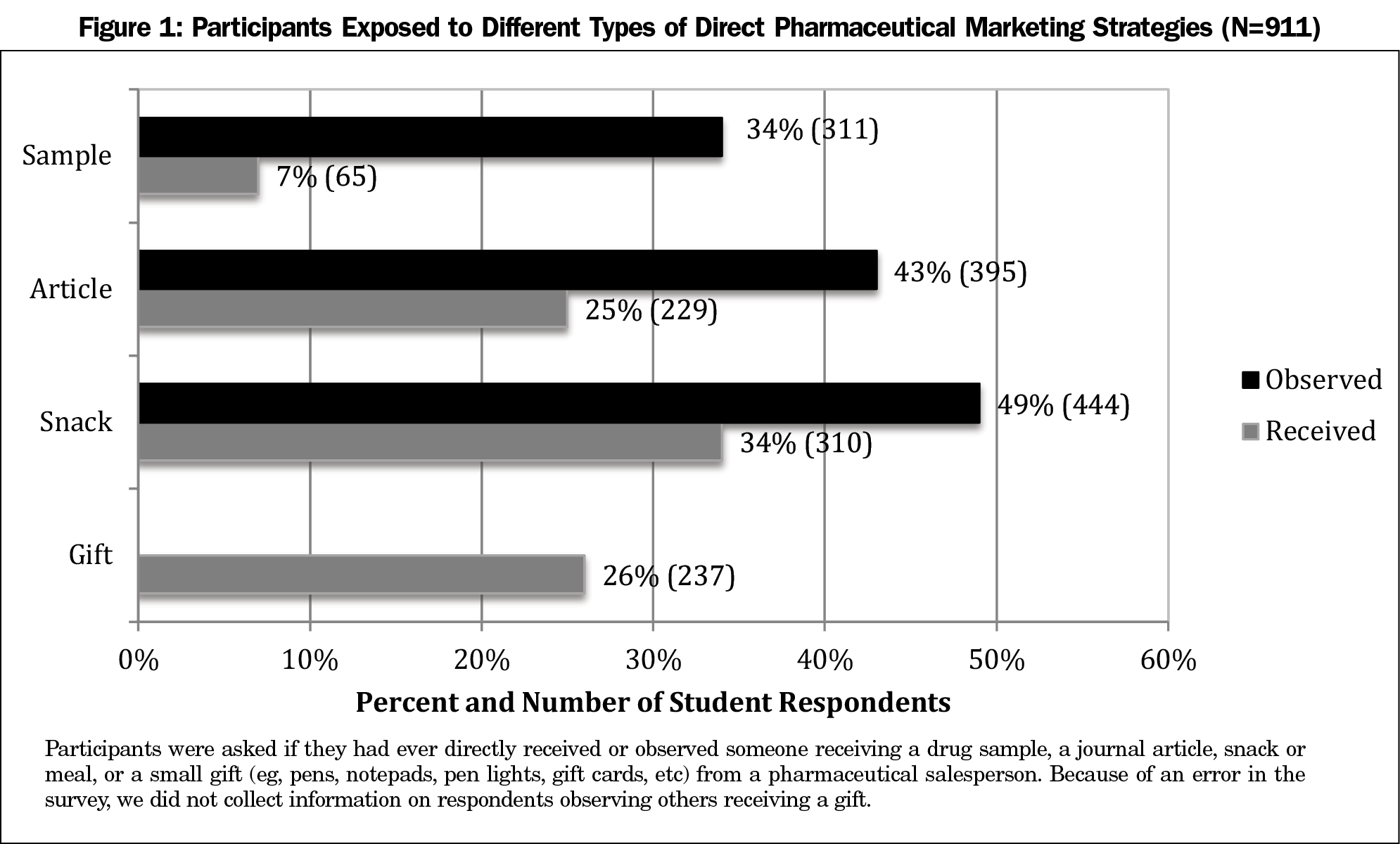
Figure 1 shows the percentage of participants who reported exposure to pharmaceutical marketing from a salesperson or observing others being similarly exposed to direct pharmaceutical marketing. Students observed others receiving a small meal or snack, articles, and samples more than they themselves received these items. The most common exposure was for students to personally receive (34%, 310) or to observe someone receiving (49%, 444) a small meal or snack.
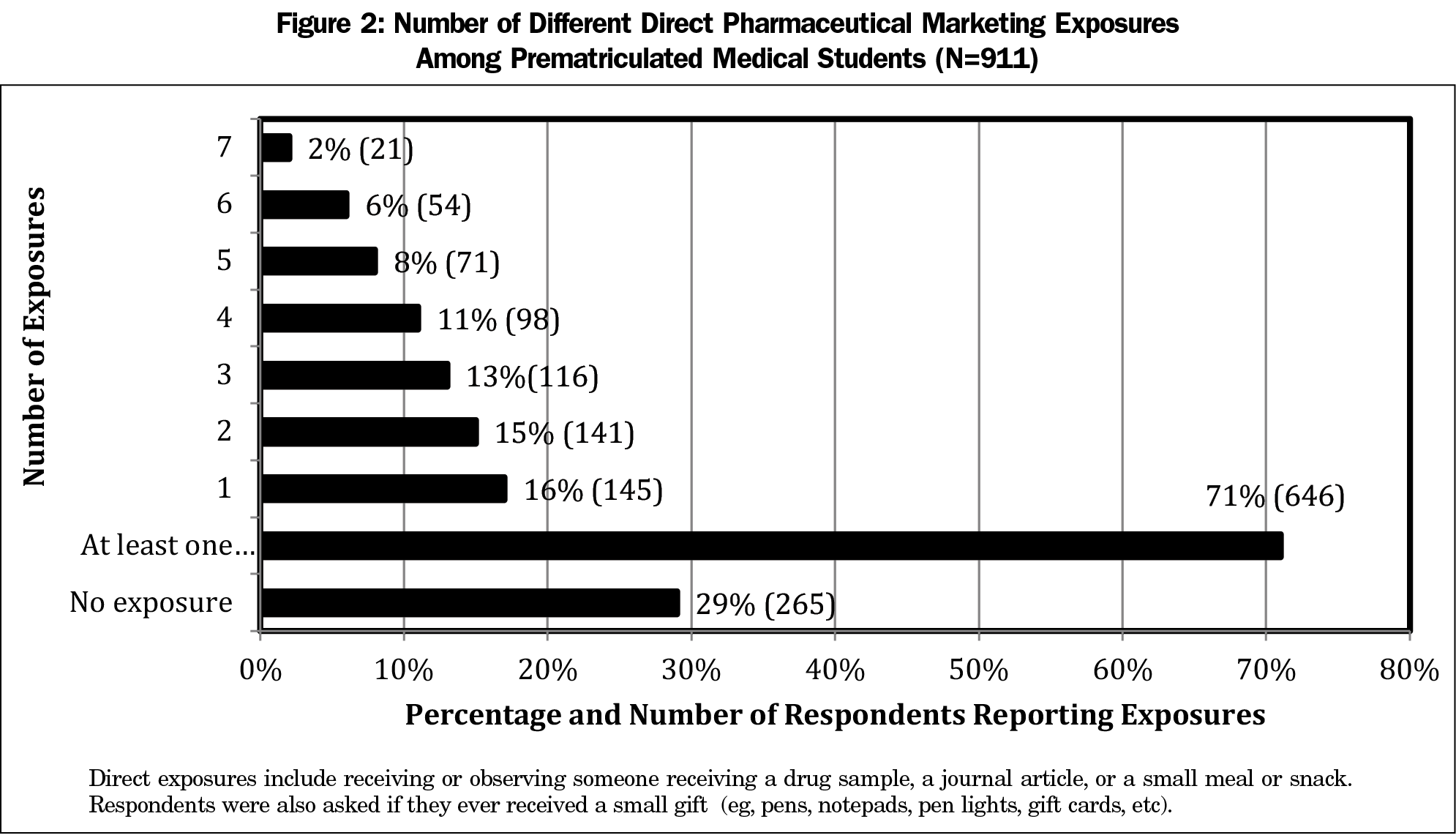
The majority of students (71%, 646) received or observed someone receiving at least one type of pharmaceutical marketing exposure (Figure 2). In addition, 40% (360) of the participants reported three or more different types of direct marketing exposures. Only 29% (265) reported no direct marketing exposures. The Northeast represented the highest percentage of pharmaceutical marketing exposure (82%, 54), followed by the South at 77% (232), the West at 70% (137) and the Midwest at 64% (220).
Among the prematriculated students who reported a pharmaceutical marketing interaction with a sales representative, a majority reported that their exposures took place while either shadowing (54%, 346) or during previous employment (50%, 323). Figure 3 illustrates the context in which premedical students encountered direct pharmaceutical marketing.
This survey of premedical students matriculating into medical school examined the prevalence and context of prior direct pharmaceutical marketing interactions. Most participants in the study entered medical school already having been exposed to some type of direct marketing from pharmaceutical salespeople. We also found that shadowing experiences were the most common place where premedical students encountered direct pharmaceutical marketing.
If premedical students form a favorable impression of pharmaceutical companies based on marketing exposure delivered to themselves or their mentors prior to matriculation, they may sustain or welcome such exposures later in their training and be more vulnerable to non-evidence-based influence on their prescribing.28-33 Given that many students appear to be exposed to this type of pharmaceutical marketing before they arrive at medical school, the current interventions may be too late or insufficient to counteract earlier, more favorable premedical student impressions about pharmaceutical marketing.
With medical schools requiring applicants to gain experience in medicine and medical practice, most students seek volunteer, shadowing, or work experiences (such as working as a medical assistant or scribe) in a medical setting. The expectation on the part of medical schools for this medical experience places applicants at risk of being influenced by direct-to-physician pharmaceutical marketing before they can be advised of the risks and potential harms of such marketing. This raises the issue of whether medical schools and premedical organizations should be providing guidance or making educational materials available to premedical students in order to increase their awareness of the known downsides of physician/pharmaceutical company interactions.
This study has a number of strengths. It was a national study that included premedical participation from all four US census regions. The survey questions were based on our review of the literature and our research team’s previous research experience in this area.27-33
The study also has important limitations. Fourteen of 188 identified US public medical schools elected to participate in this study (7% response rate). Further, among participating universities, we experienced a relatively low response rate (40%) from the prematriculating students. The study design also excluded those premedical students who did not matriculate into a public medical school. Although nonparticipant students’ experiences with pharmaceutical marketing may differ from the experiences of our participants in a nonrandom fashion, it is unclear whether this is actually the case, and whether it would increase or decrease the overall incidence of exposure in our sample.
Another limitation is the possibility of recall bias. The survey asked respondents to report past exposures. Accordingly, there is likelihood that not all responses accurately detailed all pharmaceutical sales encounters. However, this study reports on accepted medical students before they enter medical school, potentially minimizing recall bias seen in previous studies. In addition, the small number of respondents from the Northeast region limits the generalizability of any conclusions that might be drawn in comparison to other regions. Finally, there was no comparison group, nor was there representation of matriculants from most public medical schools or any private medical schools. Nonetheless, for those responding institutions, premedical student exposure to direct pharmaceutical marketing was observed or received by the majority of respondents.
We recommend that medical schools consider ways to mitigate the potential unintended consequences of premedical shadowing requirements. One approach is to offer educational material on admissions websites that inform premedical students about conflicts of interest that can arise from direct pharmaceutical marketing exposure. In addition, medical schools should also consider providing conflict-of-interest training as early as orientation. Medical associations can also play a role by educating their members about this issue and discouraging pharmaceutical salespeople visits, particularly when learners are in their clinics. Finally, premedical organizations should also provide conflict-of-interest training to their members.
Acknowledgments
Financial Support: Financial support for this study was proved by the Society of Teachers of Family Medicine Foundation Project Fund-Humanities and Ethics Collaborative.
The authors thank the Society of Teachers of Family Medicine for financial support. They also thank the 14 medical schools that assisted with the coordination of the survey and the 911 premedical participants.
Presentations: Prematriculation exposure to pharmaceutical marketing: Too much, too soon? Society of Teachers of Family Medicine’s Medical Student Education Conference, Works in Progress, Austin, Texas, February 3, 2018.
References
- Fischer PM, Schwartz MP, Richards JW Jr, Goldstein AO, Rojas TH. Brand logo recognition by children aged 3 to 6 years. Mickey Mouse and Old Joe the Camel. JAMA. 1991;266(22):3145-3148. https://doi.org/10.1001/jama.1991.03470220061027
- Lieb K, Koch C. Medical students’ attitudes to and contact with the pharmaceutical industry: a survey at eight German university hospitals. Dtsch Arztebl Int. 2013;110(35-36):584-590.
- Brody H. Pharmaceutical industry financial support for medical education: benefit, or undue influence? J Law Med Ethics. 2009;37(3):451-460, 396. https://doi.org/10.1111/j.1748-720X.2009.00406.x
- Campbell EG, Rao SR, DesRoches CM, et al. Physician professionalism and changes in physician-industry relationships from 2004 to 2009. Arch Intern Med. 2010;170(20):1820-1826. https://doi.org/10.1001/archinternmed.2010.383
- Zipkin DA, Steinman MA. Interactions between pharmaceutical representatives and doctors in training. A thematic review. J Gen Intern Med. 2005;20(8):777-786. https://doi.org/10.1111/j.1525-1497.2005.0134.x
- May CD. Selling drugs by “educating” physicians. J Med Educ. 1961;36(1):1-23.
- Katz D, Caplan AL, Merz JF. All gifts large and small: toward an understanding of the ethics of pharmaceutical industry gift-giving. Am J Bioeth. 2010;10(10):11-17. https://doi.org/10.1080/15265161.2010.519226
- Cialdini RB. Influence: The Psychology of Persuasion. New York: Quill William Morrow; 1993.
- Wazana A. Physicians and the pharmaceutical industry: is a gift ever just a gift? JAMA. 2000;283(3):373-380. https://doi.org/10.1001/jama.283.3.373
- Chren MM, Landefeld CS. Physicians’ behavior and their interactions with drug companies. A controlled study of physicians who requested additions to a hospital drug formulary. JAMA. 1994;271(9):684-689. https://doi.org/10.1001/jama.1994.03510330062035
- Symm B, Averitt M, Forjuoh SN, Preece C. Effects of using free sample medications on the prescribing practices of family physicians. J Am Board Fam Med. 2006;19(5):443-449. https://doi.org/10.3122/jabfm.19.5.443
- Campbell EG, Rao SR, DesRoches CM, et al. Physician professionalism and changes in physician-industry relationships from 2004 to 2009. Arch Intern Med. 2010;170(20):1820-1826. https://doi.org/10.1001/archinternmed.2010.383
- Hartung DM, Evans D, Haxby DG, Kraemer DF, Andeen G, Fagnan LJ. Effect of drug sample removal on prescribing in a family practice clinic. Ann Fam Med. 2010;8(5):402-409. https://doi.org/10.1370/afm.1135
- Evans DV, Hartung DM, Andeen G, et al. One practice’s experiment in refusing detail rep visits. J Fam Pract. 2011;60(8):E1-E6.
- Boltri JM, Gordon ER, Vogel RL. Effect of antihypertensive samples on physician prescribing patterns. Fam Med. 2002;34(10):729-731.
- Orlowski JP, Wateska L. The effects of pharmaceutical firm enticements on physician prescribing patterns. There’s no such thing as a free lunch. Chest. 1992;102(1):270-273. https://doi.org/10.1378/chest.102.1.270
- Austad KE, Avorn J, Franklin JM, Kowal MK, Campbell EG, Kesselheim AS. Changing interactions between physician trainees and the pharmaceutical industry: a national survey. J Gen Intern Med. 2013;28(8):1064-1071. https://doi.org/10.1007/s11606-013-2361-0
- Fugh-Berman A, Brown SR, Trippett R, et al. Closing the door on pharma? A national survey of family medicine residencies regarding industry interactions. Acad Med. 2011;86(5):649-654. https://doi.org/10.1097/ACM.0b013e318212e8d4
- Brown SR, Evans DV, Fugh-Berman A. Pharmaceutical industry interactions in family medicine residencies decreased between 2008 and 2013: a CERA study. Fam Med. 2015;47(4):279-282.
- Pew Foundation. Persuading the prescribers: Pharmaceutical industry's marketing and its influence on physicians and patients. https://www.pewtrusts.org/en/research-and-analysis/fact-sheets/2013/11/11/persuading-the-prescribers-pharmaceutical-industry-marketing-and-its-influence-on-physicians-and-patients. Published November, 2013. Accessed April 15, 2019.
- Iskowitz M. GPs Harried by Reps, Oncologists Not as Much. Medical Marketing and Media (online). https://www.mmm-online.com/home/channel/gps-harried-by-reps-oncologists-not-as-much/. Published August 30, 2012. Accessed April 15, 2019.
- Brennan TA, Rothman DJ, Blank L, et al. Health industry practices that create conflicts of interest: a policy proposal for academic medical centers. JAMA. 2006;295(4):429-433. https://doi.org/10.1001/jama.295.4.429
- Coyle SL; Ethics and Human Rights Committee, American College of Physicians-American Society of Internal Medicine. Physician-industry relations. Part 2: organizational issues. Ann Intern Med. 2002;136(5):403-406. https://doi.org/10.7326/0003-4819-136-5-200203050-00015
- Lipoff JB, Grant-Kels JM. Should dermatology residents accept educational support sponsored or funded by pharmaceutical companies? J Am Acad Dermatol. 2013;68(5):854-857. https://doi.org/10.1016/j.jaad.2012.10.025
- Kowalenko T, Char D, Marco C, et al; Society for Academic Emergency Medicine Graduate Medical Education, Ethics, and Industry Relations Committees. Industry relations with emergency medicine graduate medical education programs. Acad Emerg Med. 2009;16(10):1025-1030. https://doi.org/10.1111/j.1553-2712.2009.00539.x
- Association of American Medical Colleges. Industry funding of medical education, report of the AAMC task force. https://members.aamc.org/eweb/upload/Industry%20Funding%20of%20Medical%20Education.pdf. Published 2008. Accessed January 7, 2015.
- Hodges LE, Arora VM, Humphrey HJ, Reddy ST. Premedical students’ exposure to the pharmaceutical industry’s marketing practices. Acad Med. 2013;88(2):265-268. https://doi.org/10.1097/ACM.0b013e31827bfbce
- Barfett J, Lanting B, Lee J, Lee M, Ng V, Simkhovitch P. Pharmaceutical marketing to medical students: the student perspective. McGill J Med. 2004;8(1):21-27.
- Fein EH, Vermillion ML, Uijtdehaage SH. Pre-Clinical Medical Students’ Exposure to and Attitudes Toward Pharmaceutical Industry Marketing. Med Educ Online. 2007;12(1):4465.
- Evans DV, Keys T, Desnick L, A Andrilla CH, Bienz D, Rosenblatt R. Big pharma on the farm: students are exposed to pharmaceutical marketing more often in rural clinics. Fam Med. 2016;48(7):561-564.
- McCormick BB, Tomlinson G, Brill-Edwards P, Detsky AS. Effect of restricting contact between pharmaceutical company representatives and internal medicine residents on posttraining attitudes and behavior. JAMA. 2001;286(16):1994-1999. https://doi.org/10.1001/jama.286.16.1994
- Epstein AJ, Busch SH, Busch AB, Asch DA, Barry CL. Does exposure to conflict of interest policies in psychiatry residency affect antidepressant prescribing? Med Care. 2013;51(2):199-203. https://doi.org/10.1097/MLR.0b013e318277eb19
- Sierles FS, Kessler KH, Mintz M, et al. Changes in medical students’ exposure to and attitudes about drug company interactions from 2003 to 2012: a multi-institutional follow-up survey. Acad Med. 2015;90(8):1137-1146. https://doi.org/10.1097/ACM.0000000000000686



There are no comments for this article.